Advertisement
Advertisement
March 7, 2025
Basis Medical’s Seclusion Catheter for Superficial Vein Reflux Receives FDA Clearance
 March 7, 2025—Basis Medical recently announced that its Seclusion catheter for the treatment of superficial vein reflux has received FDA 510(k) clearance.
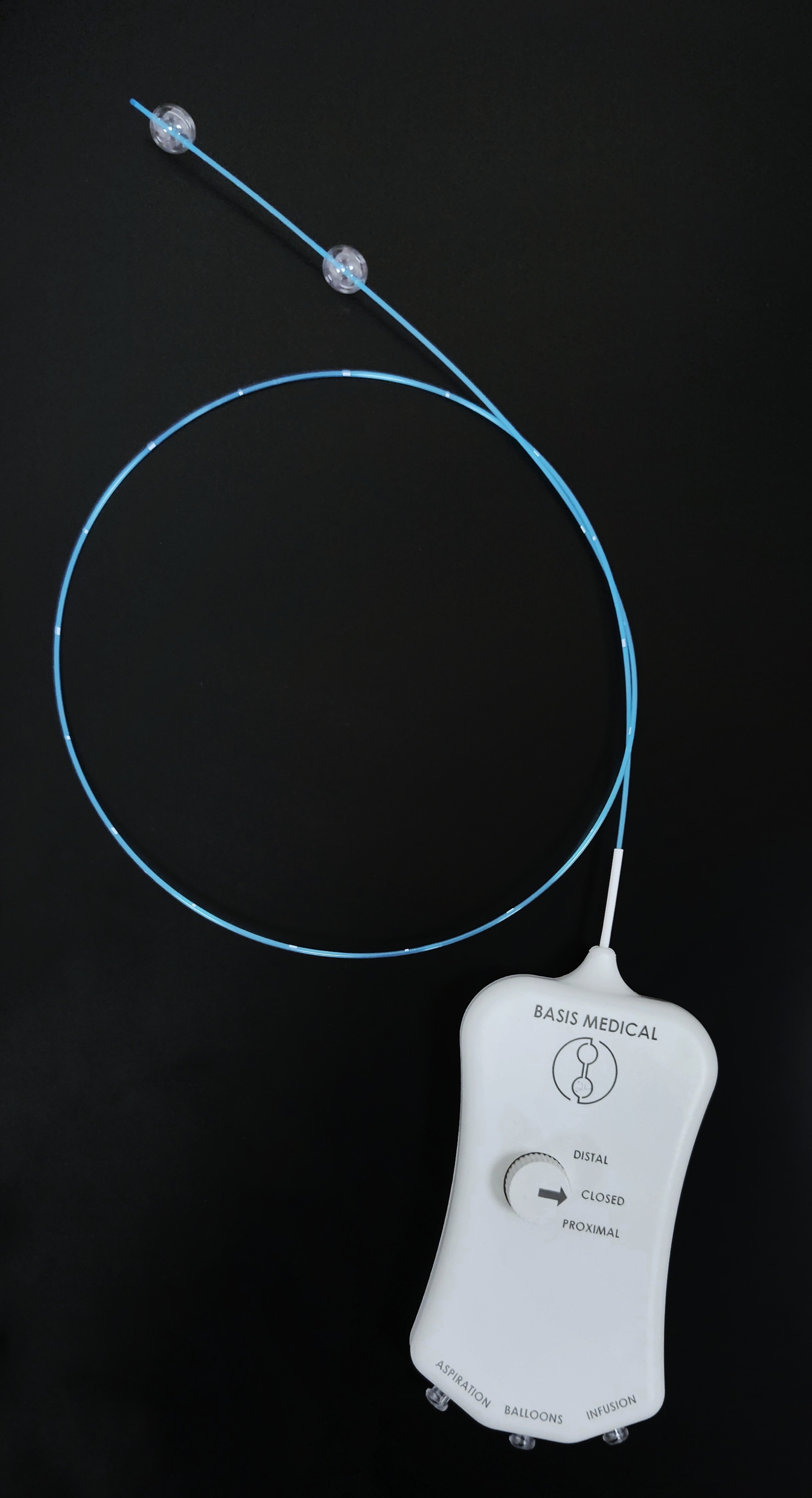
March 7, 2025—Basis Medical recently announced that its Seclusion catheter for the treatment of superficial vein reflux has received FDA 510(k) clearance.
According to the company, Seclusion is an endovenous chemical ablation device designed to provide a safe, comfortable, and clinically effective solution for patients with superficial vein reflux and chronic venous insufficiency.
Basis Medical stated that the Seclusion catheter is a dual-balloon system that temporarily isolates specific segments of diseased blood vessels, allowing for precise treatment with an FDA-approved sclerosant. This minimally invasive approach eliminates the need for thermal methods and tumescent anesthesia, noted the company.
“This FDA 510(k) clearance is a pivotal moment for Basis Medical,” commented CEO Tomas Levinton in the company’s press release. “The Seclusion catheter’s unique design enhances procedural efficiency while reducing risk and pain, benefiting both patients and physicians. We are excited to bring this technology to the market and continue our mission of delivering solutions that elevate patient care.”
Advertisement
Advertisement
