Advertisement
Advertisement
October 20, 2022
Endologix Alto Abdominal Stent Graft’s 12-Month ELEVATE IDE Trial Results Published
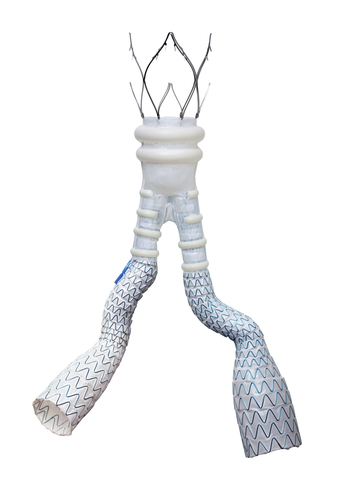 October 20, 2022—Endologix LLC announced the publication of clinical outcomes of the ELEVATE investigational device exemption (IDE) study demonstrating the safety and effectiveness at 1-year postprocedure of the company’s Alto abdominal stent graft system in endovascular aneurysm repair (EVAR) of abdominal aortic aneurysms (AAAs).
October 20, 2022—Endologix LLC announced the publication of clinical outcomes of the ELEVATE investigational device exemption (IDE) study demonstrating the safety and effectiveness at 1-year postprocedure of the company’s Alto abdominal stent graft system in endovascular aneurysm repair (EVAR) of abdominal aortic aneurysms (AAAs).
Data from the ELEVATE study were used in a premarket approval supplement to the FDA in 2019. The company announced FDA approval of the Alto system in March 2020. The company noted that the Alto device utilizes a low-profile delivery system and features the company’s adaptive sealing technology, which molds in situ to the patient’s specific aortic neck anatomy.
The findings were published by Principal Investigator Sean P. Lyden, MD, et al online ahead of print in the Journal of Vascular Surgery. Dr. Lyden will also present the study data at the VEITHsymposium, which will be held November 15-19, 2022, in New York, New York. Dr. Lyden is Chairman of the Department of Vascular Surgery at the Cleveland Clinic in Cleveland, Ohio.
According to the company, the ELEVATE IDE study enrolled 75 patients at 16 centers in the United States between March 2017 and February 2018. Commencement of enrollment was announced in March 2017.
The ELEVATE study included patients with infrarenal AAAs with neck diameters of 16 to 30 mm at 7 mm below the lowest renal artery. Patients were followed for 30 days, 6 months, and 1 year by clinical evaluation, CT, and abdominal x-ray imaging.
The company reported that the 12-month results included a treatment success rate of 96.7% at 12 months, with all-cause mortality of 4% and no AAA-related mortality. The major adverse event rate at 30 days was 5.3%.
The study investigators concluded that the Alto stent graft is safe and effective in treating AAA with appropriate anatomy at 1 year, advised the company.
Matt Thompson, MD, President and CEO of Endologix, commented in the company press release, “The publication of results of the ELEVATE study represents a foundational block of the evidence that we are building for our Alto product line.”
Dr. Thompson continued, “To further expand this evidence base, we have recently begun enrolling patients in the JAGUAR randomized controlled trial, which is a randomized comparative study examining the hypothesis that the Alto endograft will have superior durability to competitive endografts secondary to a significant reduction in post-EVAR aortic neck dilatation.”
He added, “We believe that Alto plays a significant role in the management of AAA based on its durability, low profile, and broadest patient applicability within EVAR.”
Advertisement
Advertisement
