Advertisement
Advertisement
July 18, 2023
Endologix Detour System Introduced in United States With Targeted Market Release
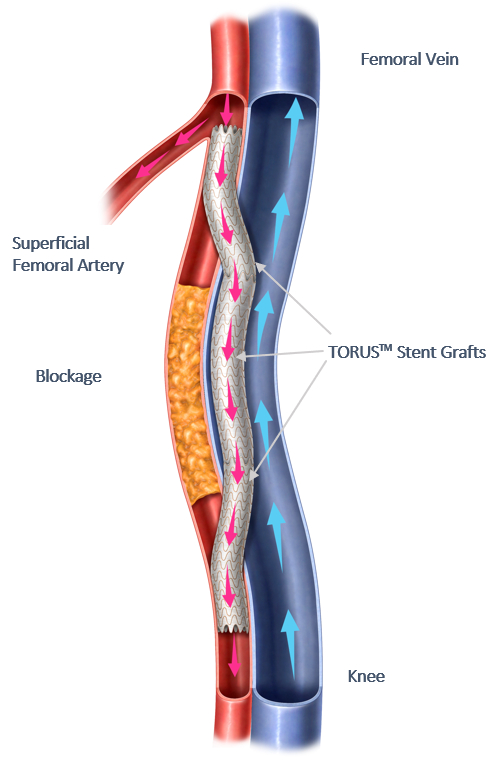 July 18, 2023—Endologix LLC announced commencement of a targeted market release of the company’s Detour system for percutaneous transmural arterial bypass (PTAB) in the United States. The first patients were treated with PTAB using Detour since the system received FDA approval, which Endologix announced on June 8, 2023.
July 18, 2023—Endologix LLC announced commencement of a targeted market release of the company’s Detour system for percutaneous transmural arterial bypass (PTAB) in the United States. The first patients were treated with PTAB using Detour since the system received FDA approval, which Endologix announced on June 8, 2023.
For the rollout, Endologix collaborated with two health care systems where PTAB using the Detour system was first performed: the Cleveland Clinic’s Sydell and Arnold Miller Family Heart, Vascular & Thoracic Institute in Cleveland, Ohio, and the Salinas Valley Health Medical Center in Salinas, California.
According to the company, PTAB with the Detour system treats complex peripheral artery disease by enabling physicians to bypass lesions in the superficial femoral artery using stents routed through the femoral vein to restore blood flow to the leg. This approach is effective for patients with long lesions (20-46 cm in length), those who have already undergone failed endovascular procedures, or those who may be suboptimal candidates for open surgical bypass.
The Detour system is indicated for percutaneous revascularization in patients with symptomatic femoropopliteal lesions from 200 to 460 mm in length with chronic total occlusions (100-425 mm) or diffuse stenosis > 70% who may be considered suboptimal candidates for surgical or alternative endovascular treatments. The Detour system, or any of its components, is not for use in the coronary and cerebral vasculature, advised the company.
Advertisement
Advertisement
