Advertisement
Advertisement
September 24, 2025
Jupiter Endovascular’s Vertex Catheter Receives FDA Clearance
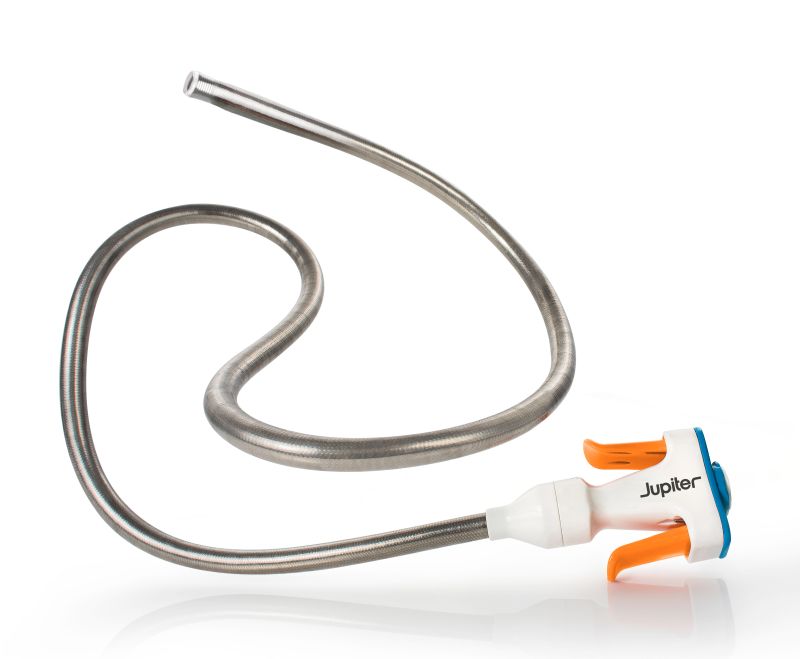 September 24, 2025—Jupiter Endovascular, Inc. announced that its Vertex catheter has received FDA 510(k) clearance for the insertion of endovascular devices.
September 24, 2025—Jupiter Endovascular, Inc. announced that its Vertex catheter has received FDA 510(k) clearance for the insertion of endovascular devices.
The Vertex intravascular device features the company’s Endoportal Control platform with TFX—transforming fixation—technology that allows it to shift from flexible to firm on demand for stability and control, stated the company.
Jupiter Endovascular’s announcement, which was posted on its LinkedIn media page, includes the link to the FDA database listing for the clearance.
In October 2024, the company announced the commencement of the SPIRARE II United States pivotal trial of the Vertex system with Endoportal Control for pulmonary embolectomy to treat acute pulmonary embolism in an endovascular procedure.
Advertisement
Advertisement
