Advertisement
Advertisement
June 14, 2024
Medical Templates’ Cube Navigator Software Receives FDA Clearance
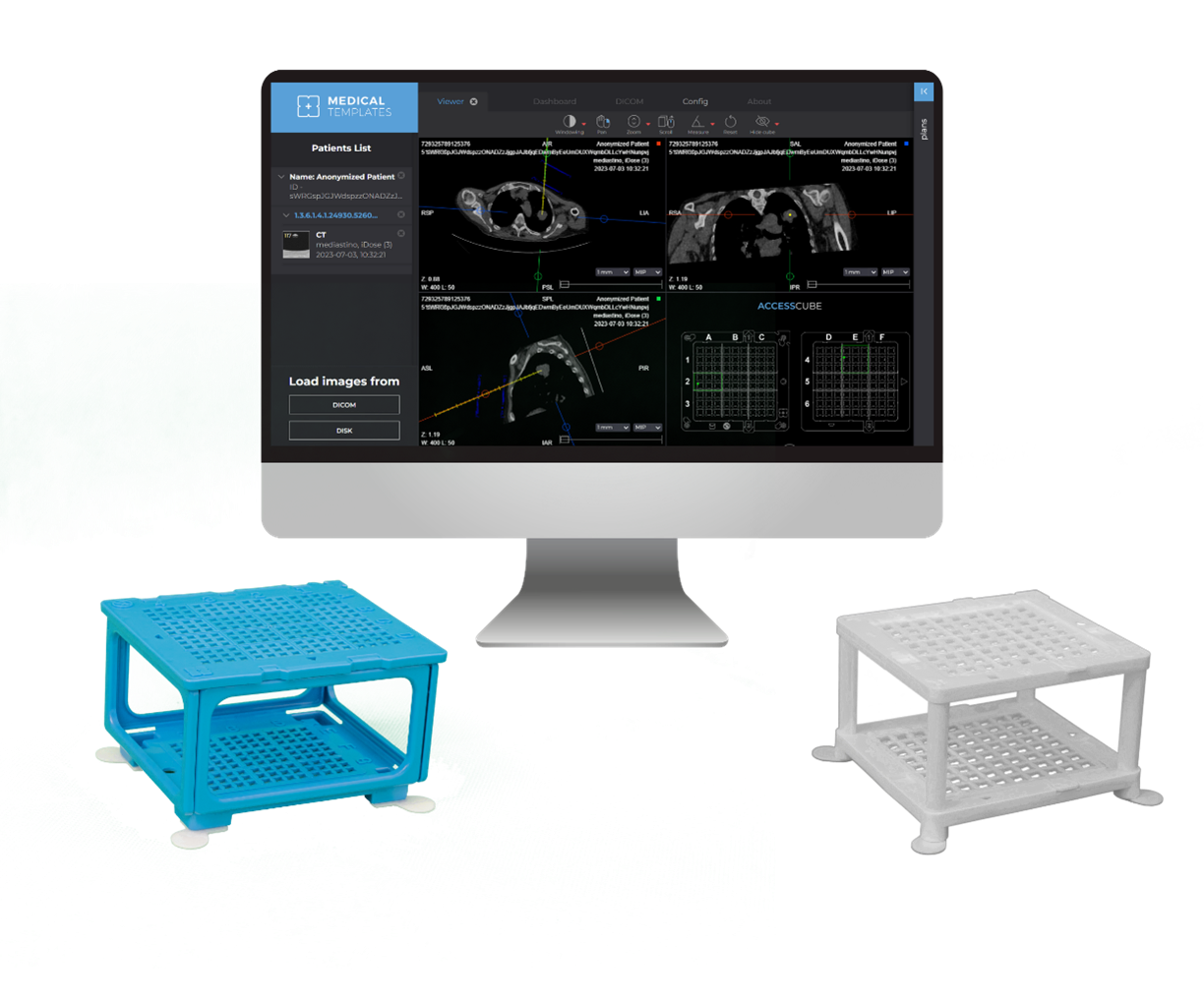 June 14, 2024—Medical Templates, a Switzerland-based health startup, announced recently that it received FDA clearance for the company’s Cube Navigator software for planning of percutaneous CT-guided interventions.
June 14, 2024—Medical Templates, a Switzerland-based health startup, announced recently that it received FDA clearance for the company’s Cube Navigator software for planning of percutaneous CT-guided interventions.
The Cube Navigation system’s computer vision-enabled software pairs with Medical Templates’ hardware—a needle guide in the form of a cube—as an alternative to the free-hand method (FHM) or capital-intensive solutions such as robots, the company says. Previously available in Europe, the product will now be launched in the United States.
Medical Templates noted the increased importance of navigation, especially expanding treatment options in oncology.
The company cited a recent study that found that punctures performed with the Cube Navigation system compared with FHM were > 4 times closer to the target on the initial puncture (2.5 ± 1.2 mm vs 12.1 ± 7.7 mm), had significantly more accurate final needle locations (3.8 ± 1.3 mm vs 6.7 ± 4.5 mm; P = .004), and were significantly faster (263.1 ± 84.4 s vs 411.2 ± 141.0 s; P < .001).
The findings were published by Lorenz Krammer et al in Journal of Vascular and Interventional Radiology (2023;34:1809-1814). Additional studies on accuracy and speed are currently being carried out in Switzerland and Germany, advised Medical Templates.
Advertisement
Advertisement
