Advertisement
Advertisement
March 20, 2023
SurgVision’s Explorer Air II Cleared by FDA for Fluorescence-Guided Oncology Procedures
March 20, 2023—SurgVision GmbH, part of the Bracco Group, announced that it has received FDA 510(k) clearance for the Explorer Air II for use with pafolacianine (Cytalux, On Target Laboratories Inc.) during intraoperative fluorescence imaging. SurgVision is a Munich, Germany–based developer of solutions for fluorescence-guided surgical and interventional oncology.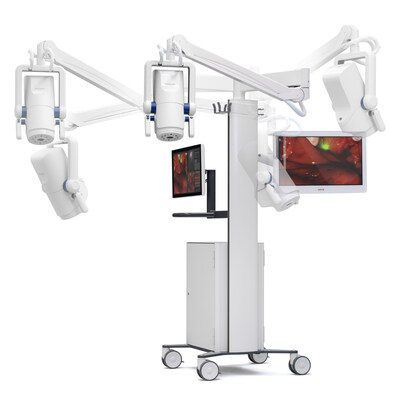
According to the company, this is the first product based on SurgVision’s next-generation technology platform. It is designed to achieve high sensitivity and imaging fidelity to meet the needs of oncological intraoperative fluorescence imaging. The system allows real-time imaging during surgery, and its prototype has been tested by academic centers for a variety of indications.
Additionally, the Explorer Air II is cleared in the United States and CE Mark approved in the European Union for visual assessment of blood flow and tissue perfusion.
The company noted that with current practice, the identification of tumors during surgery or interventional endoscopy relies on visual inspection and palpation. Tumor tissue is sometimes difficult to distinguish from healthy tissue; consequently, surgical resection of the tumor is often incomplete.
SurgVision stated its pipeline combines highly sensitive imaging solutions to visualize tumors during surgical or interventional procedures in real time, enabling sensitive and accurate detection of the tumor.
Advertisement
Advertisement
