Advertisement
Advertisement
February 26, 2026
Vena Medical’s MicroAngioscope Cleared by FDA for Use in Peripheral Vasculature
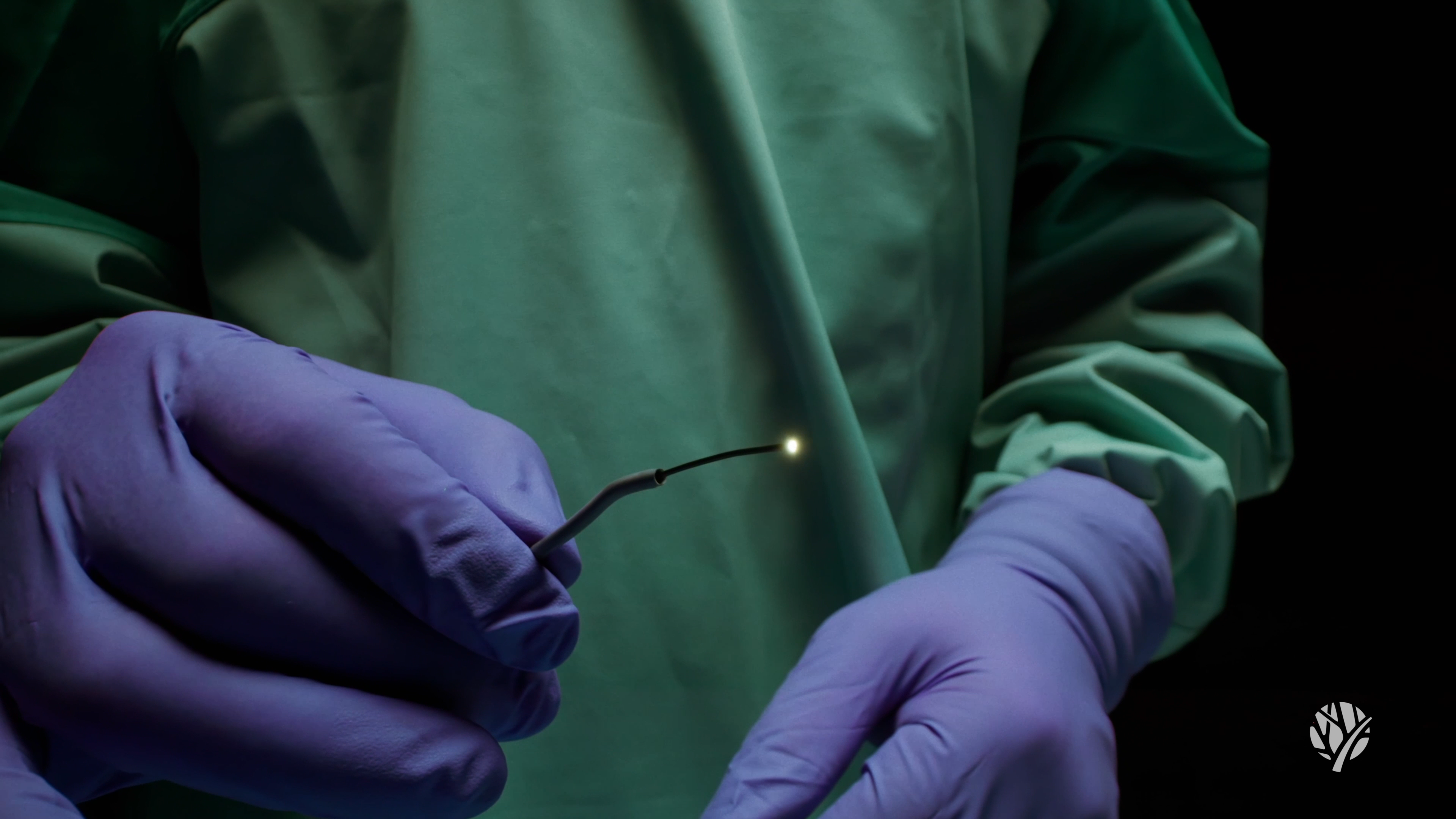
The company advised that the regulatory milestone opens the door for international exporting of its flagship technology, beginning with entry into the United States through a limited market release of the system.
According to the company, the Vena MicroAngioscope provides full-color, live imaging directly within peripheral blood vessels, enabling physicians to make more informed treatment decisions in real time. The technology has been used in Canada to treat more than 100 patients, noted Vena Medical.
To support its commercial expansion, Vena Medical has opened a $4.5 million Manufacturing Center of Excellence in Kitchener, Ontario. The investment brings the production of Vena’s microcamera technology in-house and helps fulfill the company’s commitment to keep manufacturing local, stated the press release.
Vena Medical advised that the state-of-the-art facility was supported by strategic funding from Ontario’s Life Sciences Scale-Up Fund. It was officially unveiled in a ribbon-cutting ceremony led by the Honorable Vic Fedeli, Ontario’s Minister of Economic Development, Job Creation, and Trade.
Advertisement
Advertisement
