Advertisement
Advertisement
June 30, 2025
Basis Medical’s Seclusion Catheter Used in FIH Clinical Procedures to Treat CVI
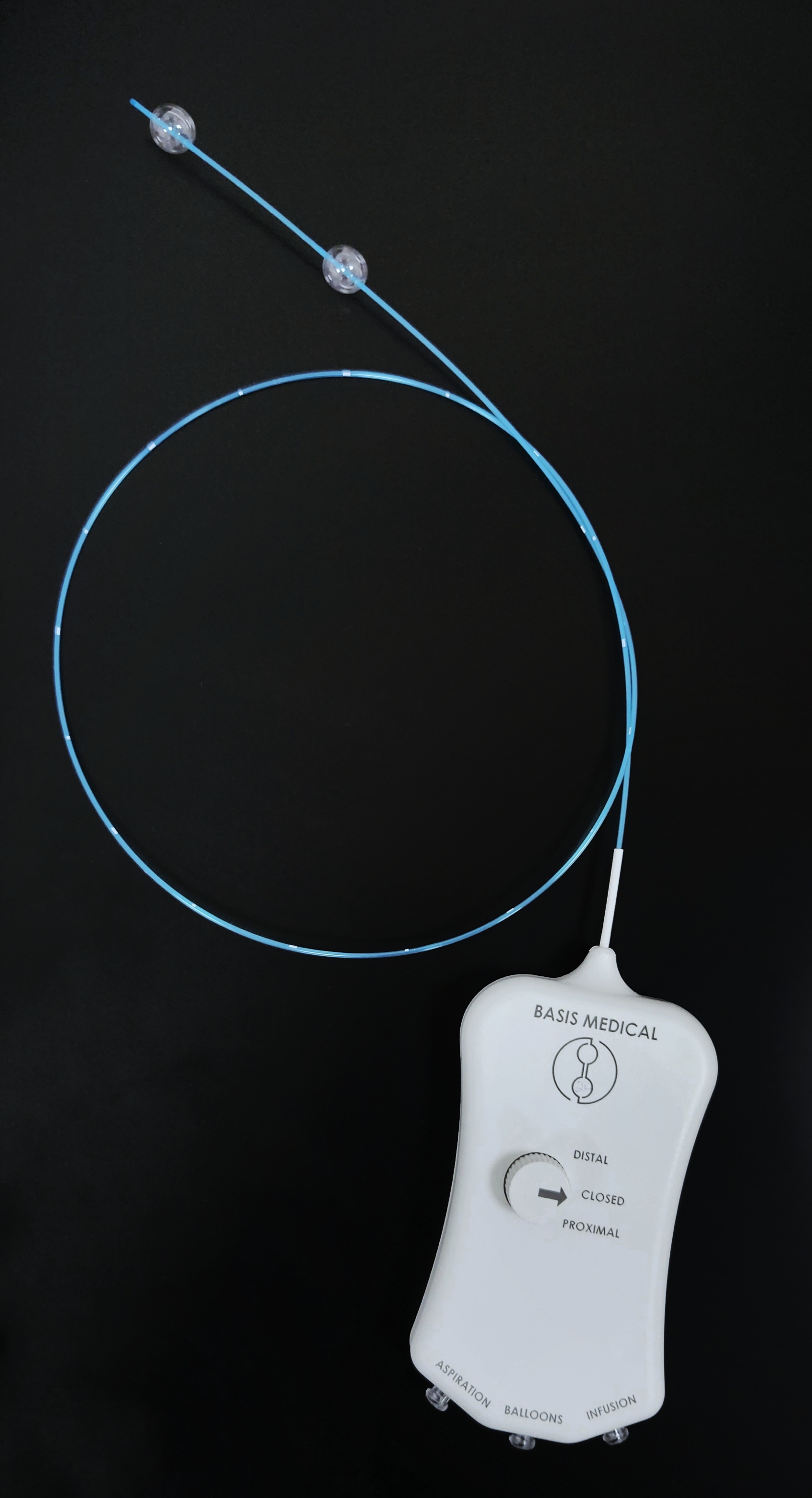 June 30, 2025—Basis Medical announced the completion of the first-in-human clinical procedures using the company’s Seclusion catheter to treat chronic venous insufficiency (CVI) caused by superficial vein reflux. In March 2025, the company announced FDA 510(k) clearance for the Seclusion device.
June 30, 2025—Basis Medical announced the completion of the first-in-human clinical procedures using the company’s Seclusion catheter to treat chronic venous insufficiency (CVI) caused by superficial vein reflux. In March 2025, the company announced FDA 510(k) clearance for the Seclusion device.
According to Basis Medical, the cases involved patients with symptomatic CVI and visible varicosities caused by reflux in the great saphenous vein. All patients tolerated the procedure well, and no complications were reported during or immediately after treatment, advised the company.
As noted in the press release, Basis Medical’s dual-balloon Seclusion catheter is designed specifically to treat superficial venous reflux and CVI in the legs. It is engineered to deliver targeted therapy to malfunctioning superficial veins, providing a nonthermal, nontumescent approach to vein ablation.
The company stated that the cases were performed by Marcos Fletcher, MD, a cardiovascular surgeon at Ciudad de Salud Hospital in Panama City, Panama.
“The Seclusion catheter represents a significant advancement in the treatment of superficial vein disease,” commented Dr. Fletcher in the company’s press release. “It delivered excellent control, precision, and patient comfort. We successfully treated target segments both above and below the knee, and the early results are highly encouraging. This technology has the potential to set a new standard in vein care.”
To support its upcoming commercial launch in the United States and advance clinical trials, Basis Medical is currently raising a $6 million Series A financing round, noted the press release.
Advertisement
Advertisement
