Advertisement
Advertisement
September 7, 2023
Endologix Detour System Granted CMS New Technology Add-On Payment
 September 7, 2023—Endologix LLC announced that the Centers for Medicare & Medicaid Services (CMS) granted a new technology add-on payment (NTAP) for the company’s Detour system for percutaneous transmural arterial bypass (PTAB) as part of the CMS FY2024 hospital inpatient prospective payment system. The company noted that an NTAP facilitates patient access to qualifying new medical technologies that substantially improve the diagnosis or treatment of Medicare beneficiaries.
September 7, 2023—Endologix LLC announced that the Centers for Medicare & Medicaid Services (CMS) granted a new technology add-on payment (NTAP) for the company’s Detour system for percutaneous transmural arterial bypass (PTAB) as part of the CMS FY2024 hospital inpatient prospective payment system. The company noted that an NTAP facilitates patient access to qualifying new medical technologies that substantially improve the diagnosis or treatment of Medicare beneficiaries.
Beginning October 1, 2023, CMS will provide hospitals with additional device reimbursement when the Detour system is used for eligible cases in the hospital inpatient setting.
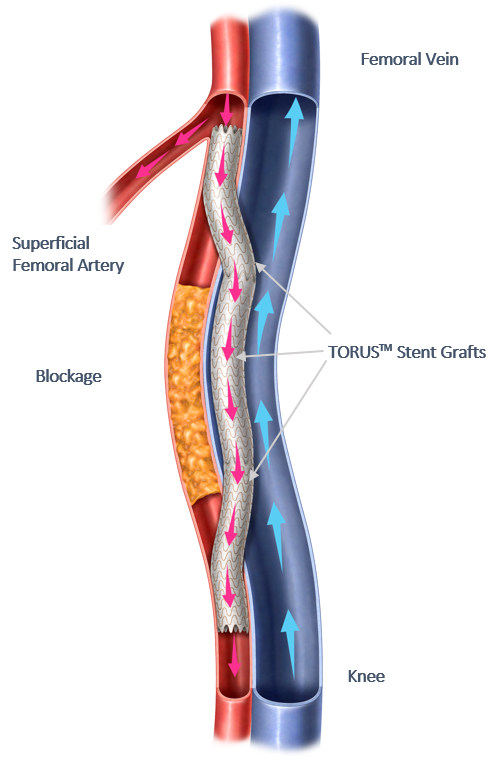
According to Endologix, PTAB with the Detour system offers a novel approach to treating complex peripheral artery disease, enabling physicians to bypass lesions in the superficial femoral artery by using conduits routed through the femoral vein via a transmural passage to restore blood flow to the leg. This approach is effective for patients with long lesions (20-46 cm in length), those who have already undergone failed endovascular procedures, or those who may be suboptimal candidates for open surgical bypass.
On July 18, 2023, Endologix announced the commencement of a targeted market release of the Detour system for PTAB in the United States. The company announced FDA approval of the device on June 8. The device received FDA Breakthrough Device designation in 2020.
Advertisement
Advertisement
